Survey for Ceratocystis fimbriata on Syngonium
Upasana Dhakal*, Chris Kadooka and Janice Y. Uchida Plant and Environmental Protection Sciences, CTAHR, University of Hawaii at Manoa, Honolulu, HI 96822
Ohia (Metrosideros polymorpha Gaud.) is one of the foundation trees of the Hawaiian rainforest ecosystem at the island of Hawaii, and makes up about 80% of the total trees in the remaining forests on the Hawaiian Islands. Ohia forest supports Hawaiian rainforest ecosystem which consist of many flora and fauna among which 90% are endemic. Ohia forest holds significant cultural and environmental importance. Since 2009-2010 ohia trees have been rapidly dying, due to a disease caused by a fungi, Ceratocystis lukuohia and Ceratocystis huliohia. They are new species in Ceratocystis fimbriata species complex. The disease is known as Rapid Ohia Death (ROD). Over 3,824 ha of ohia forest were affected in 2014 which has increased to 75,000 ha by 2017. C. fimbriata has a broad host range and is known to cause vascular wilts of woody trees and rots on non-woody plants. C. fimbriata is very close to C. lukuohia and C. huliohia and is known to cause stem and leaf rot of Syngonium in Hawaii but nothing is known about the diversity of C. fimbriata from Syngonium. There might be multiple strains and some might be pathogenic to ohia. Thus, it is important to determine if this pathogen is present on other Islands on Syngonium and determine its host range.
1. Conduct surveys on Oahu, Hawaii to determine if Ceratocystis frimbriata is present on Syngonium and where.
2. Test the pathogenicity of Isolates collected from Syngonium on Syngonium and ohia.
Figure 1: Locations surveyed on Oahu and C. fimbriata symptoms on Syngonium. 1A: GPS coordinates of the locations surveyed on Oahu. 1B: Dark lesions on the basal node and leaf base and rotting of the plant from the base.
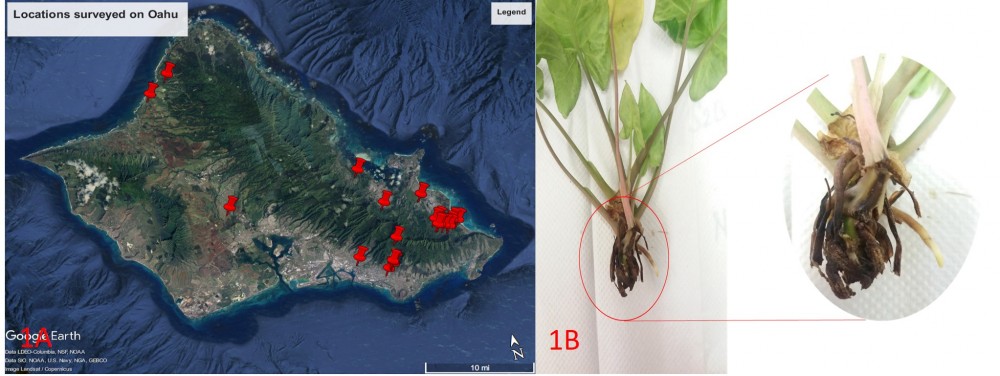
Figure 2: Syngonium podophyllum before and after inoculation. 2A: Healthy plant before inoculation. 2B: 3 days post inoculation (Dpi). 2C: 7 Dpi, 2D: Severe wilt and death of leaves at 14 Dpi.

Figure 3: Ohia before and after inoculation. 3A: Healthy ohia plant before inoculation. 3B: 57 days post inoculation.

Surveys of the island of Oahu and the island of Hawaii were conducted. Washed samples were dissected into smaller pieces, surface disinfected with 10% Clorox and plated on water agar or incubated under moist conditions. Single hyphal tips of the mycelium growing out from the infected samples or ascospore masses protruding out of perithecia were transferred to10% vegetable juice agar. Twenty two different locations in Oahu including, commercial nurseries, botanical gardens and retail garden centers were surveyed (Figure 2A). C. fimbriata was not isolated from any samples collected on Oahu. Most common symptom was the rotting of the aerial root tips. Fusarium species was the most commonly isolated fungus. Other fungi were also recovered, less frequently. Out of three nurseries surveyed in Big island, samples from two of them were found to be infected with C. fimbriata. Pathogenicity of C. fimbriata isolate 3401 and 3459 was tested on Syngonium and ohia. 20 µl of a spore suspension at 1x 107 endoconidia per ml sterile distilled water were pipetted into the holes drilled halfway across on ohia stem. Wounds on one set of ohia plants were covered with parafilm while in other set wounds were uncovered. Unwounded Syngonium plants were thoroughly sprayed with spore suspension at 1x 106 endoconidia per ml sterile distilled water. Each treatment was replicated 3 times.
During the survey, no symptoms typical to C. fimbriata were observed in most of the diseases. Black discoloration of the node was observed a few times, but C. fimbriata was not isolated. C. fimbriata from Syngonium was pathogenic to Syngonium (Figure 2). Ohia plants are still under observation. Fungus isolated from different samples during the surveys have been purified and tubed for long term storage. Pathogenicity will be tested whenever host are available.
