Phase I: Refining Vegetative Propagation Techniques for Federally Listed Bonamia menziesii (Convolvulaceae) on Kauaʻi
Rebekah Magers, Ashly Trask, Natalia Tangalin, Randy Umetsu

 Vegetative cuttings were taken over the course of six months in NTBGʻs Conservation and Horticulture Center and at two field locations on Kauaʻi. Five different IBA hormones were used to initiate root growth: Hormex #3, #8, #45, Clone X, and PhytoGro2. There were attempts to keep similar sample sizes for all hormones used during this project, however, nursery and wild cutting quantities varied due to younger plants in the nursery in comparison to older, larger Bonamia menziesii found in the wild.
Vegetative cuttings were taken over the course of six months in NTBGʻs Conservation and Horticulture Center and at two field locations on Kauaʻi. Five different IBA hormones were used to initiate root growth: Hormex #3, #8, #45, Clone X, and PhytoGro2. There were attempts to keep similar sample sizes for all hormones used during this project, however, nursery and wild cutting quantities varied due to younger plants in the nursery in comparison to older, larger Bonamia menziesii found in the wild.
+ New Cuttings
All cuttings were put into an identical substrate and mist house environment for the propagation period. Substrate mix used was a 4:1 of extra-large perlite to vermiculite. The mist house is an open air structure with 20 seconds of mist every 30 minutes for 24 hours, 7 days a week. Rain was still able to penetrate through the shade cloth. Nursery stock cuttings were taken in the late morning to afternoon. Cuttings were made with a minmum of 3 nodes per cutting or 3-4 inches in length. Cuttings were immediately wounded along one side of the stem on the cut end, dipped in hormone, inserted into substrate then placed in the mist house. Wild cuttings were taken on two separate field trips and a lapse of approximately 19-63 hours passed before they were recut and followed the same procedure. Wild cuttings were kept in an oversized cooler bag while in the field and refrigerated before being brought to the nursery.
Cuttings with 3-9 nodes (dependent on internode length) followed the same procedure before being stuck in substrate: top cut was directly above leaf node, bottom cut was directly below leaf node where last leaf was removed (Fig. 1), wounds were about an inch in length, made by scraping pruners along the stem on one side (Fig. 2). Cuttings were then dipped in hormone and a pencil was used to displace substrate before inserting cutting. All leaves were kept on for nursery cuttings but leaves were cut from wild cuttings to minimize transpiration while in the field.

Fig. 1
 Fig. 2
Fig. 2
 Photo of labeled cuttings in mist house.
Photo of labeled cuttings in mist house.
+ Observations
Observations were made approximately every 7-14 days in the nursery starting April 27, 2018. Weather (temperature, cloud cover, UV index, humidity), start and end time, plant accession number, hormone, position of cutting (vertical or horizontal), and stem type were all recorded along with visual observations of callusing, leaf drop, stem color, new growth and root growth. The best method decided for observing root growth was by gently scooping each propagule out of the substrate with a spoon (Fig. 3). Another set of wild cuttings from June 7, 2018 were left undisturbed.
 Fig. 3
Fig. 3

The purpose of this project was to learn what concentration of rooting hormone produced the most effective rooting of B. menziesii. This project helped lay the groundwork for effective propagation while building up NTBG's ex situ collection for future projects and out plantings.
Five IBA hormones were used to initiate rooting in nursery and wild cuttings: Hormex #3, #8, #45, Clone X and PhytoGro2. Hormex #45 was not used after April 13, 2018 because of observed low success rates early on. Hormex #3 was trialed in its place for future cuttings. Previous work at NTBG with wild cuttings resulted in lower rooting success compared to nursery cuttings. This project showed little difference between wild and nursery cuttings’ rooting success (Fig. 4).
 Fig. 4
Fig. 4
The graph represents the percentage of successful rooting per hormone used on wild and nursery cuttings that were scooped and observed until 6/29/18 (Graph 1).
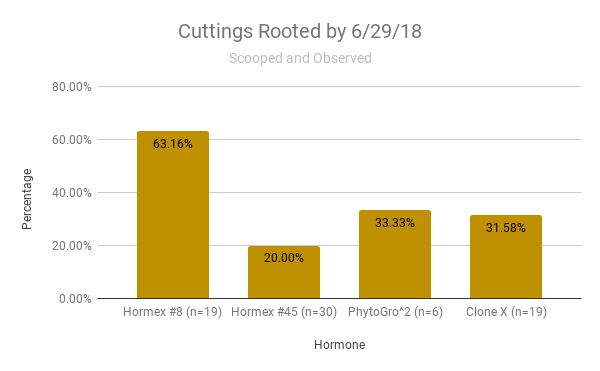 Graph 1.
Graph 1.
The graph represents the percentage of successful rooting per hormone on wild cuttings that were left undisturbed for 35 days once they were put into substrate (Graph 2).
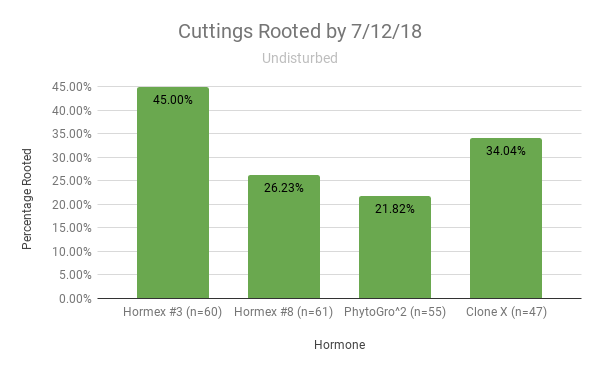 Graph 2.
Graph 2.
 Photos of root formation on B. menziesii cuttings.
Photos of root formation on B. menziesii cuttings.


+ Abstract
Bonamia menziesii, is a U.S. Federally listed endangered species of endemic Hawaiian vine. Historically it was found on all major Hawaiian Islands, except Niʻihau and Kahoʻolawe. It is now extirpated on Molokaʻi. Populations on Kauaʻi are only loosely defined and it’s estimated there are less than a couple hundred individuals in Hawaiʻi (Perlman, et al.) (Map 1). National Tropical Botanical Gardens’ (NTBG) Living Collections Department is hoping to expand their ex situ population of this woody vine and build a nursery stock for future research and restoration projects. Since being federally listed in 1994, little attention has been paid to B. menziesii. There is much room for research in regards to its reproductive biology, pollinators, and vegetative propagation techniques. In the past, it was observed that wild cuttings had a significantly smaller rooting percentage than those taken in the nursery and quickly put in the mist house (Trask). In Phase I cumulative trials were carried out with both nursery and wild collected propagules to refine combinations of hormones with different stem cutting types. Various intervals of callusing, nodule and root formation were observed and recorded. Observation of root growth, even with careful extraction methods, can be disruptive to new root growth. This poster will explain the methodology, successes and challenges for Phase I of refining B. menziesii propagation techniques. Phase II of this project, to be completed summer of 2018, will be attempting hand pollination techniques and subsequent seed collection on established ex situ collections.
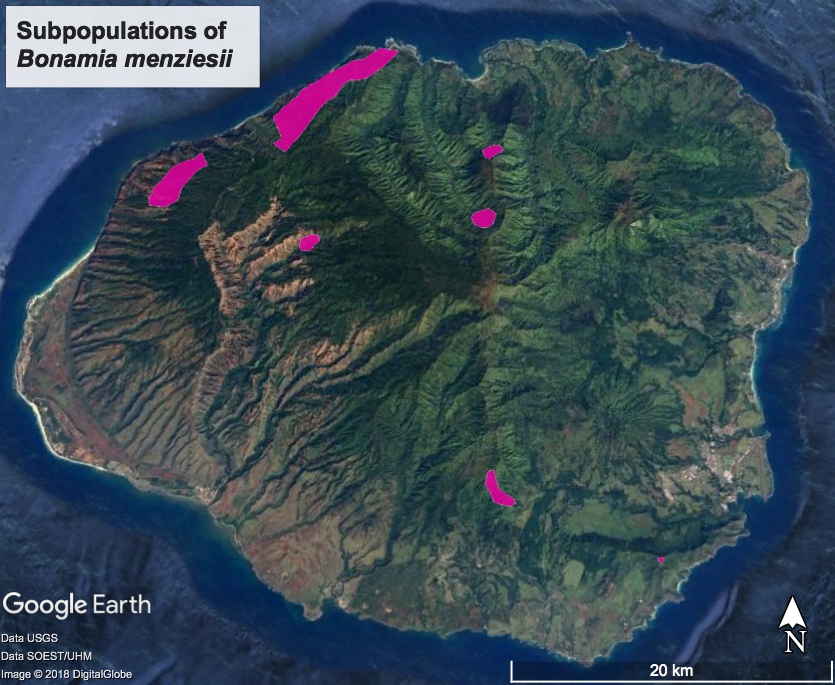
Map 1. Preliminary map of B. menziesii subpopulations on Kauaʻi, ca. 20 colonies with 100± total individuals.
 Photo of finding B. menziesii in the wild.
Photo of finding B. menziesii in the wild.

One of the major threats to B. menziesii in the wild, aside from ungulate habitat degradation, drought, land slides and flooding, is lack of reproduction (USFWS). Phase II will focus on pollination and seed storage for future projects.
+ Hand Pollination
In the summer of 2018, interns continued to work on B. menziesii by focusing on self and cross breeding through hand pollination in the nursery. Hand pollination may be necessary as populations decline due to loss of native pollinators and reduced diversity within small wild populations. No flowers were noticed on any plants while in the field during Phase I of this project. In the nursery plants were self or cross pollinated and marked with distinct flagging tape. Finally, on June 6, 2018 a seed pod was achieved through cross pollination on an older nursery B. menziesii (Fig. 5).
 Fig. 5
Fig. 5
+ Pollinators
B. menziesii has a small tubular flower with white petals covered in beige pubescence (Fig. 6). The shape and color of its flower suggests that a small moth may be an important pollinator in its life cycle (Buchmann). Also observed on B. menziesii in the field was an unidentified weevil (Fig. 7). As priority research topics of The Hawaiʻi Plant Conservation Network (HPCN), life biology and pollinators need more time and attention to gain a better understanding of this endemic.
 Fig.6
Fig.6
 Fig. 7
Fig. 7

Throughout this project there were a few challenges in using both nursery and wild cuttings.
+ Setup & Observation Methods
One of the main challenges was deciding if cuttings should be removed and observed or left undisturbed in the substrate. From prior experience, wild cuttings resulted in lower rooting success and so disturbing each cutting was thought to be intrusive. It was decided to use a spoon to carefully scoop out each cutting and observe initial callusing and onset of rooting. Using this method there was an observed average of 39 days for initial root formation.
 Photo of undisturbed wild cuttings of B. menziesii using Hormex #3, observed after 35 days on 7/12/18.
Photo of undisturbed wild cuttings of B. menziesii using Hormex #3, observed after 35 days on 7/12/18.
+ Time
Another challenge is timing. Nursery cuttings are able to be cut, wounded and dipped in hormone immediately, all at one location. Even while kept cool, wild cuttings had to endure hours of travel from the field to the nursery before being put into the substrate. Leaves on wild cuttings were also cut to reduce transpiration during travel.
 Photo of cutting leaves to reduce transpiration.
Photo of cutting leaves to reduce transpiration.
Bruegmann, M.M. & Caraway, V. 2003. Bonamia menziesii. The IUCN Red List of Threatened Species 2003: e.T44129A10862572. http://dx.doi.org/10.2305/IUCN.UK.2003.RLTS.T44129A10862572.en. Accessed on 04 July 2018.
Environmental Conservation Online Services (ECOS). US Fish and Wildlife Services. https://ecos.fws.gov/ecp0/profile/speciesProfile?sId=2503. Accessed on 04 July 2018.
Laukahi: The Hawaiʻi Plant Conservation Network. p. 30. laukahi.org/wp-content/uploads/2017/12/Laukahi-Research-Suggestions-List.pdf Access June 20, 2018.
Buchmann, Stephen, et al. Selecting Plants for Pollinators: A Regional Guide for farmers, land managers, and gardeners. The Pollinator Partnership. https://www.fws.gov/Pollinators/pdfs/HawaiianIsland.EcoRegGuide.FINAL.hi-res.pdf
Trask, Ashly. Personal Interview. 29 June 2018
Tangalin, Natalia. Personal Interview. 6 June 2018
Perlman, Steve; Tangalin, Natalia; Wood, Ken. "Re: Bonamia Menziesii Project." Message to Rebekah Magers. 8 July 2018. E-mail.
