Changes in obligate coral-feeding reef fish populations after mass coral bleaching at Lisianski Island, Northwestern Hawaiian Islands
Roseanna Lee, University of Hawaii at Hilo, Department of Marine Science


Fishes within Coral Reefs
The abundance and distribution of reef fishes can be affected by the availability of live coral (Booth & Beretta 200). An estimated 30% of all 15,000 describe marine fishes live in coral reefs (Moberg & Folke 1999). Many of these fish may be important bioindicators of healthy reefs, including obligate corallivores that require coral polyps as food (Friedlander & Parrish 1998).
Coral Bleaching
Coral bleaching, the loss of Symbiodinium spp. in coral polyps and the breakdown of their symbiotic relationship, is a response to environmental stressors, such as changes in temperature, salinity, and light (Coles & Jokiel 1978). Symbiodinium are microscopic algae that photosynthesize, providing a food source for hermatypic corals that fulfills the majority of their energy requirements (Muscatine et al. 1981). Long-term stress and the lack of Symbiodinium replenishment can lead to mortality in corals (Wilkinson et al. 1999). Mass coral bleaching events are attributed to rising ocean temperatures, which have been increasing in frequency and severity since the 1980s (Coles & Jokiel 1978).
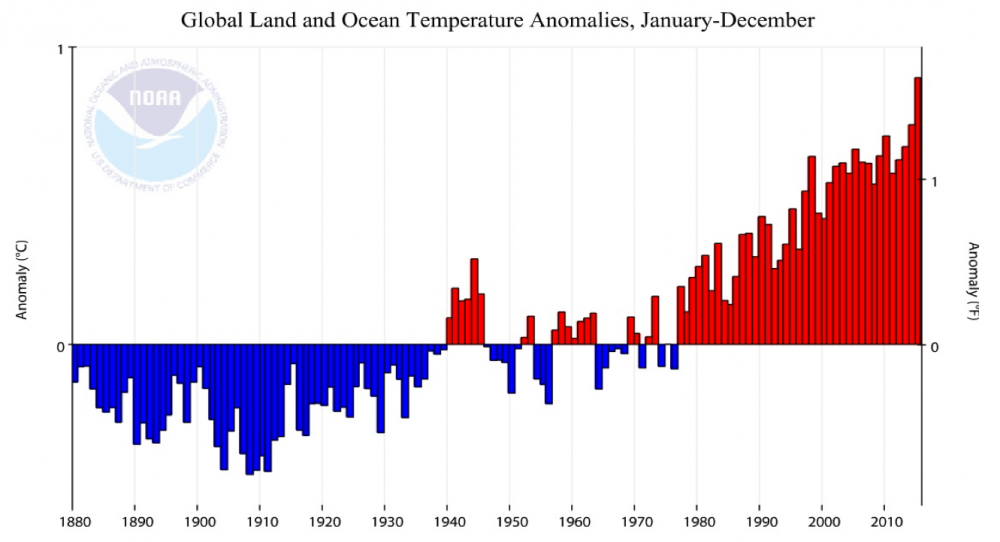
This study examined the effects of coral bleaching on the obligate coral-feeding reef fish in the Northwestern Hawaiian Islands, in particular, the effects of the 2014 mass coral bleaching event.

Background
The Northwestern Hawaiian Islands experienced their sixth, and largest, recorded coral bleaching event in 2014 (Couch et al. 2017). There was an average of 41% coral bleaching at Lisianski island with one site that experienced 99% corals bleached.

Location
This study was conducted in the Papahānaumokuākea Marine National Monument at Lisianski Island. Papahānaumokuākea Marine National Monument is the second largest marine protected area in the at 1,508,870 km2 and surrounds ten islands and atolls, known as the Northwestern Hawaiian Islands.
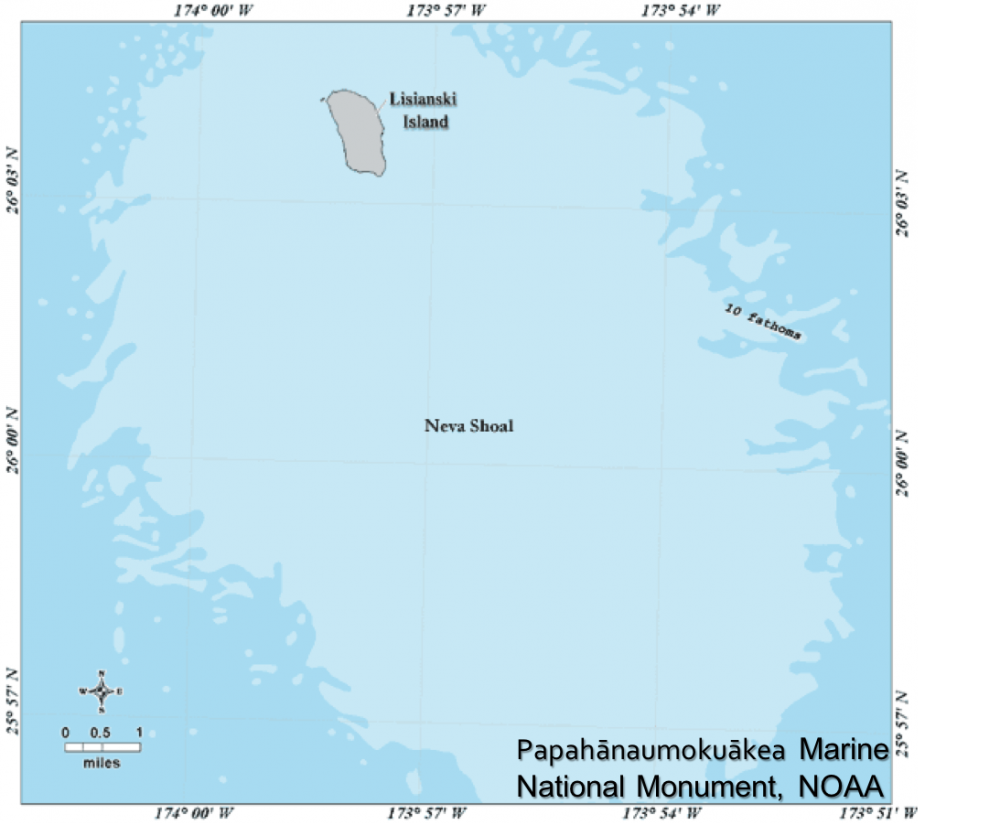
Data Collection
Diver-based surveys were conducted to assess fish assemblages and coral bleaching during a research expedition to the Northwestern Hawaiian Islands aboard the National Oceanic and Atmospheric Administration (NOAA) ship, Hiʽialakai, in 2014 (HA-14-04; August 2014) as part of the Pacific Reef Assessment and Monitoring Project through the NOAA Coral Reef Ecosystem Division (CRED).
Fish Surveys
During surveys, two divers counted, sized, and identified all fishes observed within a 15m diameter cylinder, along a 30m transect, using the Rapid Ecological Assessment (REA) new Stationary Point Count (nSPC) method (Ayotte et al. 2015).

Benthic Analysis
Subsequent to the fish surveys, divers took still photos of the benthos (Ayotte et al. 2015). At every meter mark along the 30m transect, a diver took a digital photograph of the benthic substratum by holding the camera 1m above the hard bottom, using a 1m sizing shaft for distance, with the transect line centered in the photo.
Benthic photos were visually analyzed for estimated percent cover. Each segment was analyzed along nine parameters; % healthy tissue, % bleached tissue, % diseased tissue, % recently dead coral, % turf algae, % macroalgae, % crustose coralline algae (CCA), % sand, and % other. The percentages for all photos along a given transect were averaged to give an average benthic composition for one transect.

Statistical Analysis
To test the hypothesis that coral bleaching has a negative effect on the abundance of corallivorous fishes, a series of regressions were run. First, to assess the preliminary data, a regression between hard coral cover and corallivorous fish abundance was evaluated to study the effects of percent coral cover on fish populations. Additional regressions were run to determine the relationship between coral bleaching and the abundance of fish in the surveyed area.
To show spatial and temporal changes of corallivorous fish in association to live coral cover, where live coral is defined by the presence of coral polyps, regardless of the condition, a regression was run between total coral cover and fish abundance. A significant positive relationship (R2 = 0.3435, p = 0.007) was seen between these two parameters (Fig. 1).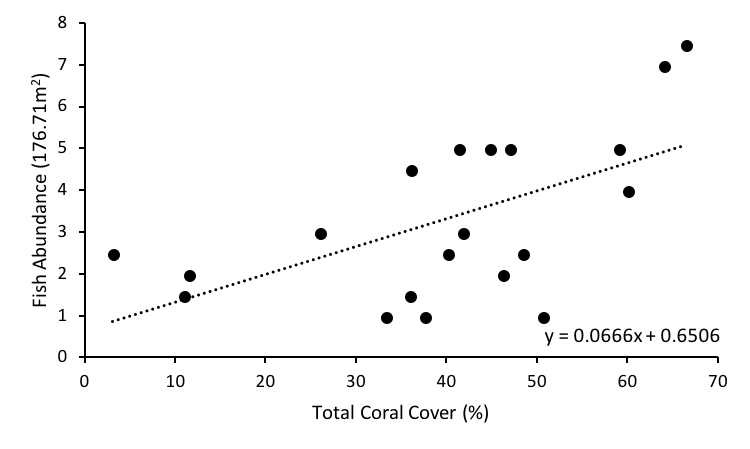
Fig. 1: Regression between total coral cover and corallivorous fish abundance at Lisianski Island, HI, USA, August 2014
The linear regression model to test the hypothesis that coral bleaching has a negative relationship with all observed corallivorous fish abundance shows a slight, positive relation (R2 = 0.1717, p = 0.069) between the two parameters, rejecting our hypothesis that coral bleaching has a negative association to corallivorous fish abundance (Fig. 2).
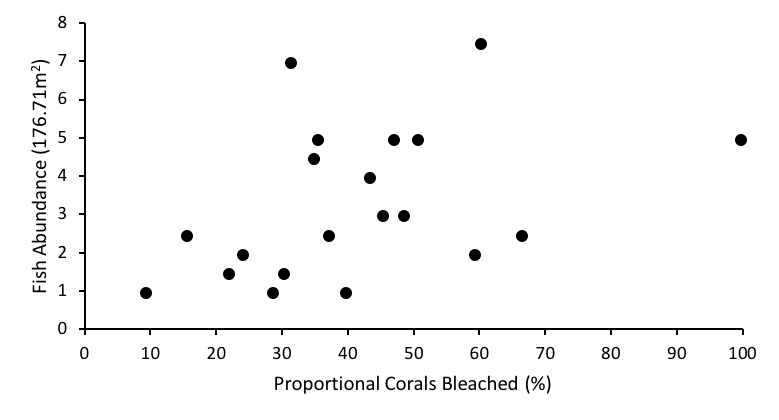
Fig. 2: Regression between proportional coral bleached and corallivorous fish abundance at Lisianski Island, HI, USA, August 2014
Spatial variation by species differs significantly by corallivorous fish species. To ensure that variability within the linear regression model between proportional coral bleached and fish abundance was not due to variability in spatial distribution by species, additional regressions were run by species against coral bleaching (Fig. 3). None of the following cases resulted in a negative relationship between fish abundance and coral bleaching. Though the significance in the relationships differed between species, all three cases show slight, positive relationships (a. R2 = 0.0525, p = 0.331, b. R2 = 0.0237, p = 0.517, c. R2 = 0.1365, p = 0.109) between fish abundance and coral bleaching.
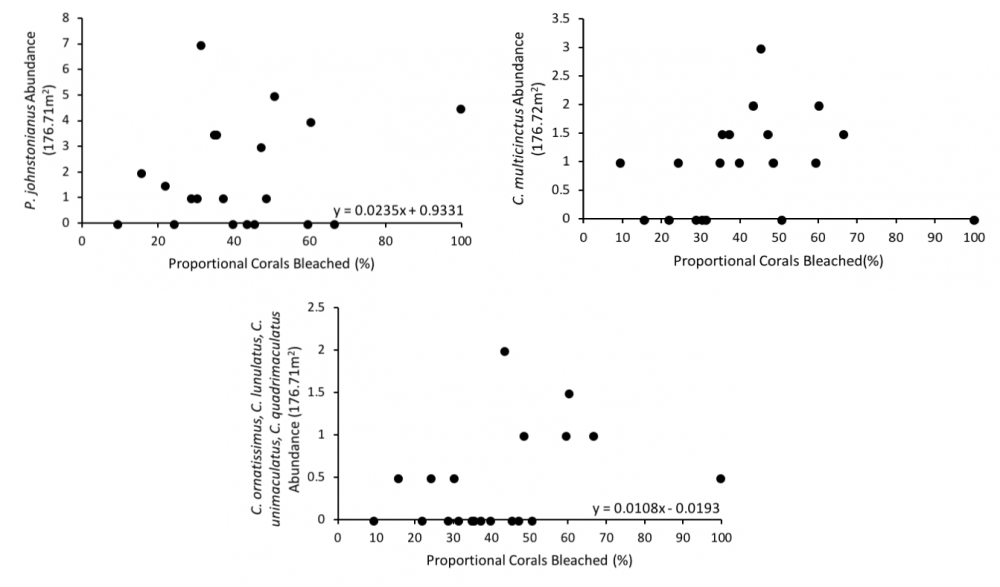
Fig. 3: Regressions between proportional corals bleached and corallivorous fish abundance by species at Lisianski Island, HI, USA, August 2014
1) This study supports the strong relation between corals and coral-feeding fishes
2) Corallivorous fish abundance is not immediatly driven by the condition of the coral
-Results concure with study by Pratchett et al.
3) Previous study shows no immediate decline in population abundance, but found bleaching impacted fish health
-Fish increased consumption of unaffected corals by shifting the primary species of coral consumed
-Lower hepatocyte vacuolation in fish during bleaching years
4) Generalists and specialists may respond in different ways
-Generalists may change feeding habits
-Specialists may change primary coral consumed
5) The abundance of corallivorous fishes increases with coral cover
6) Corallivorous fish abundance is not immediately driven by the condition of the coral cover
-Both bleached and healthy tissue have a similar relationship to fish abundance
7) The impacts of coral bleaching does not have immediate ramifications on the abundance coral feeding fishes
8) Continued monitoring of specific fish populations after bleaching events will allow us to further determine the long term effects of coral bleaching on corallivorous fish populations

A special thank you to my thesis advisor, Corinne Kane, and thesis instructors, Karla McDermid and Marta deMaintenon. I thank Randall Kosaki, Brian Hauk, Lisa Parr, and the Marine Option Program for your support in my expedition to the Northwestern Hawaiian Islands through the NOAA/MOP partner-internship. I also thank Atsuko Fukunaga, Bernardo Vergas-Angel, Timothy Tricas, Timothy Grabowski, and John Burns for analytical and statistical support. None of this research would have been possible without the support of the NOAA Ship, Hiʽialakai crew.

Literature Cited
1) Ayotte P, McCouy K, Heenan A, Willians I, Zamzow J (2015) Coral reef ecosystem program standard operating procedure: data collection for rapid ecological assessment fish surveys. Pacific Islands Fish Sci Cent, Natl Mar Fish Serv, NOAA, Admin Rep H- 15-07
2) Booth DJ, Beretta GA (2002) Changes in a fish assemblage after a coral bleaching event. Mar Ecol Prog Ser 245:2015-212
3) Coles SL, Jokiel PL (1978) Synergistic effects of temperature, salinity and light on the hermatypic coral Montipora verrucosa. Mar Biol 49(3):187–95
4) Couch CS, Burns JHR, Liu G, Steward K, Gutlay TN, Kenyon JC, Eakin CM, Kosaki RK (2017) Mass coral bleaching due to unprecedented marine heatwave in Papahānaumokuākea Marine National Monument (Northwestern Hawaiian Islands). PLoS ONE 12(9):1-27
5) Friedlander AM, Parrish JD (1998) Habitat characteristics affecting fish assemblages on Hawaiian coral reef. J Exp Mar Biol Ecol 224:1–30
6) Moberg F, Folke C (1999) Ecological goods and services of coral reef ecosystems. Ecol Econ 29:215–233
7) Muscatine L, McCloskey L, Marian R (1981) Estimating the daily contribution of carbon from zooxanthellae to coral animal respiration. Oceanography 26:601–611
8) Pratchett MS, Wilson SK, Berumen ML, McCormick MI (2004) “Sublethal effects of coral bleaching on an obligate coral feeding butterflyfish”. Coral Reefs 23:352–356
9) Wilkinson C, Linden O, Cesar H, Hodgson G, Rubens J, Strong AE (1999) Ecological and socioeconomic impacts of 1998 coral mortality in the Indian Ocean: An ENSO impact and a warning of future change? Ambio 28(2):188–96
